When your immune system is turned down-whether from steroids, chemotherapy, or transplant drugs-you don’t just get sick more often. You get sick in ways most people never even hear about. The bugs that creep into your body aren’t the usual suspects. They’re the ones that normally don’t bother healthy people. And they don’t act like normal infections either. No fever. No pus. No redness. Just quiet, creeping damage that can kill before you realize you’re in trouble.
What Makes These Infections So Dangerous?
Think of your immune system like a security team. In a healthy person, it spots invaders fast, raises the alarm, and calls in backup. In someone on immunosuppressants, that team is half-asleep. Some members are gone. Others are tied up with paperwork. So the bad guys walk right in-and they’re not just any bad guys. They’re the ones that only show up when security is down.Take Pneumocystis jirovecii. It’s everywhere. You breathe it in all the time. But in healthy people, it’s harmless. In someone with low T-cells-like after a transplant or on long-term steroids-it turns into pneumonia. And not the kind with cough and chills. Often, it just shows up as mild shortness of breath, or nothing at all. In one study of kids before stem cell transplants, 23% had this infection with zero symptoms. By the time they felt bad, it was already too late.
That’s the pattern: no warning signs. No fever. No elevated white blood cells. That’s why doctors can’t wait for classic symptoms. They have to test proactively. Bronchoscopy with lung fluid samples catches Pneumocystis in 92% of cases. Sputum tests? Only 65%. If you’re on immunosuppressants and you’re even a little short of breath, get checked. Don’t wait.
Unusual Organisms You Won’t Find in Normal Infection Guides
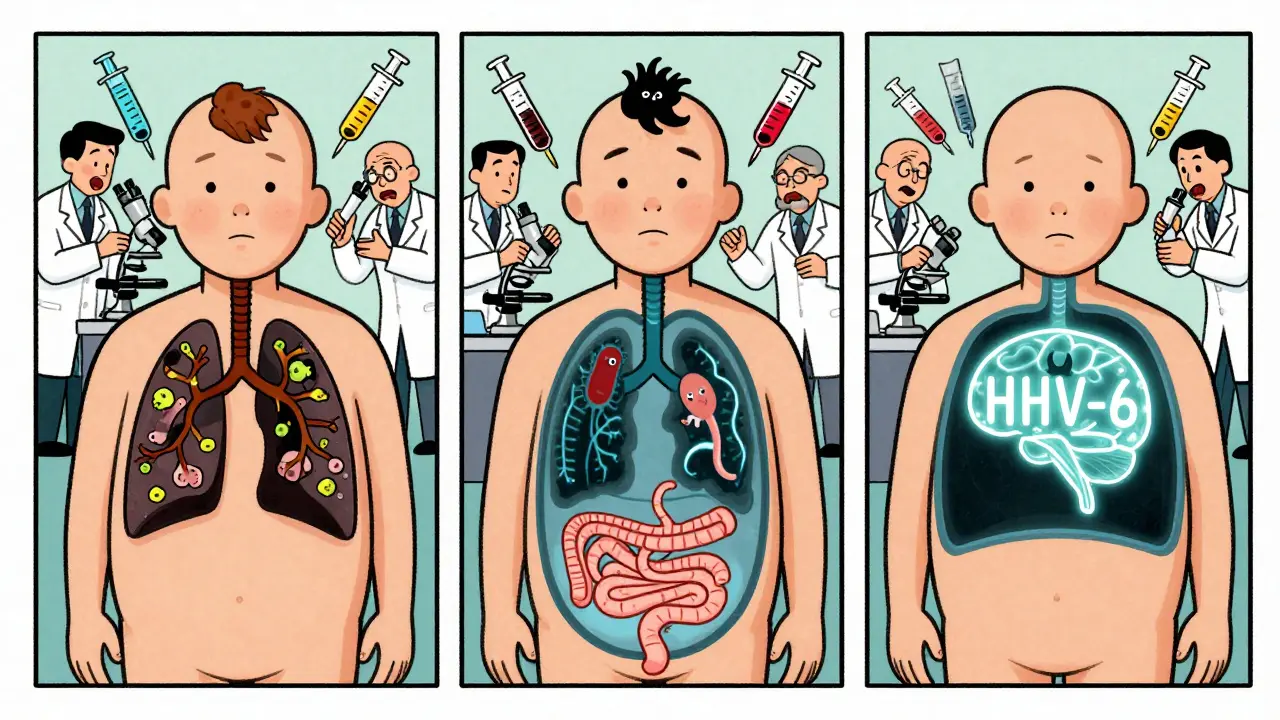
Most doctors learn about strep throat, flu, and urinary tract infections. But for immunosuppressed patients, the list of troublemakers is weirder-and more deadly.- Giardia intestinalis: This tiny parasite normally causes traveler’s diarrhea. In people with antibody deficiencies (like X-linked agammaglobulinemia), it sticks around for months. Think foul-smelling, greasy stools, bloating, and weight loss-even if you haven’t left home. It doesn’t clear on its own. Standard treatments fail 30-40% of the time in these patients.
- Aspergillus: A mold you find in soil and compost. In healthy people, it’s ignored. In someone with low neutrophils, it invades the lungs and spreads like wildfire. Mortality? Over 50%. Even with the best antifungals.
- Mycobacterium avium intracellulare: A cousin of tuberculosis. It doesn’t just hit the lungs. It spreads through the blood, infecting the liver, spleen, and bone marrow. Often mistaken for cancer.
- Human herpesvirus-6 (HHV-6): Most adults carry it. It stays quiet. But in transplant patients, it can flare and cause brain swelling, bone marrow failure, or severe skin rashes.
- Coronaviruses NL63 and HKU1: These aren’t the ones that caused the pandemic. They’re the common cold cousins. But in immunosuppressed patients, they cause severe pneumonia and last for weeks-or months.
One study of 69 immunodeficient children found that over half of all respiratory infections were caused by these unusual bugs. Pneumocystis and bacteria made up 54%. Viruses like parainfluenza, RSV, and CMV made up another third. The rest? A mix of things most labs don’t even test for routinely.
How Your Type of Immunosuppression Changes Your Risk
Not all immune suppression is the same. And your risk depends on what part of your immune system is turned off.- T-cell deficiency: This is the big one. If your T-cells are low (from steroids, anti-rejection drugs, or diseases like SCID), you’re at high risk for viruses (CMV, HHV-6, adenovirus), fungi (Pneumocystis, Aspergillus), and parasites (Toxoplasma). You’re 15 to 20 times more likely to get a viral reactivation than someone with B-cell problems.
- B-cell deficiency: Low antibodies mean trouble with bacteria like Giardia, Streptococcus pneumoniae, and Haemophilus. You’re more likely to get chronic sinus infections, pneumonia, and intestinal bugs. Your body can’t make antibodies to fight them off.
- Phagocyte defects: If your white blood cells can’t eat bacteria, you get repeated staph infections (45% of skin and bone infections), plus Gram-negative bugs like Pseudomonas and Klebsiella. Some patients even get infections from weird bugs like Flexispira, which normally live in the gut and don’t cause disease in healthy people.
- Chronic granulomatous disease: Your immune cells can’t kill certain bacteria, so they form lumps called granulomas. These show up in the liver, spleen, or lymph nodes-and they’re full of live bacteria you can’t see without a microscope.
Dr. Park’s 1967 case series showed this clearly. One patient had varicella (chickenpox) that didn’t heal for months. Another had herpes so bad it burned through skin and muscle. A third died from histoplasmosis-but it didn’t look like pneumonia. It looked like a skin infection, like erysipelas. The doctors didn’t recognize it because it didn’t match the textbook.
Why Symptoms Don’t Show Up-And What That Means for Diagnosis
You’ve been taught that fever means infection. Swelling means infection. Pain means infection. But in immunosuppressed patients? Not always.Here’s the hard truth: your body can’t mount a normal response. No fever? That’s not because you’re fine. It’s because your immune system is too weak to even try. That’s why routine screening is life-saving.
In the 2007 study, 6 out of 26 kids with confirmed infections had no symptoms at all. They were found only because doctors tested them routinely before transplant. That’s not luck. That’s protocol.
Today, guidelines recommend:
- Regular bronchoalveolar lavage (BAL) for anyone with lung symptoms-even mild ones.
- Stool testing with antigen or PCR for anyone with chronic diarrhea.
- CMV blood tests every 1-2 weeks in high-risk transplant patients.
- Imaging (CT scans) for unexplained weight loss or night sweats.
And don’t rely on blood tests alone. White blood cell counts? Often normal. CRP? Can be low. ESR? May not rise. You need direct testing-fluids, tissue samples, DNA tests-to catch these infections early.
Treatment Isn’t Just About Antibiotics
Giving the right drug isn’t enough. In immunosuppressed patients, treatment is more complex.- Giardia: Metronidazole works in healthy people-but fails 30-40% of the time in those with antibody problems. Often, you need tinidazole or nitazoxanide. Sometimes, you need both.
- Pneumocystis: Trimethoprim-sulfamethoxazole is first-line. But if you’re allergic? Pentamidine or atovaquone. And you still need steroids to calm lung inflammation-even if you’re not coughing.
- Aspergillus: Voriconazole is standard. But if it doesn’t work? Isavuconazole or amphotericin B. And even then, 50% die. That’s why early detection matters more than treatment.
- CMV: Ganciclovir or valganciclovir. But if the virus keeps coming back? You may need CMV-specific T-cell therapy. New trials show 70% of patients respond.
And here’s the catch: drugs can be toxic. Your liver and kidneys are already stressed. So dosing is tricky. You can’t just follow standard guidelines. You need to monitor blood levels. Adjust for weight. Watch for side effects.
The New Threats: COVID-19 and Beyond
The pandemic didn’t just bring SARS-CoV-2. It showed us how fragile immunosuppressed patients really are.One person with a transplant shed the virus for over 120 days. That’s more than eight times longer than a healthy person. They didn’t get sicker. They just kept infecting others. And the virus kept mutating inside them-creating new variants.
Now, coronaviruses NL63 and HKU1 are routinely tested for in transplant centers. They account for nearly 9% of respiratory infections in these patients. And they’re not going away.
Future tools are coming. Metagenomic sequencing can now find unknown bugs in lung fluid without culturing them. That’s huge. It’s how we found new viruses in patients who had no diagnosis for years.
And researchers are testing T-cell therapies that can be trained to hunt down specific viruses-without triggering graft-versus-host disease. Early results? Promising.
The Hard Truth
Despite all the advances, infection-related death rates in transplant patients still hover at 25-30%. That’s not because we don’t have drugs. It’s because we still miss the signs. We wait for fever. We wait for cough. We wait for something to look "normal." But in immunosuppressed patients, normal doesn’t exist.What saves lives isn’t a new drug. It’s suspicion. It’s testing before symptoms. It’s knowing that a little shortness of breath, a few days of diarrhea, or a vague feeling of fatigue could be the first sign of something deadly.
If you’re on immunosuppressants, don’t wait for the textbook. Ask for the test. Push for the scan. Demand the culture. Because in your case, the infection isn’t unusual because it’s rare. It’s unusual because it’s silent.
Can immunosuppressed patients get infections from common cold viruses?
Yes. Viruses like RSV, influenza, parainfluenza, adenovirus, and even the common cold coronaviruses (NL63 and HKU1) can cause severe pneumonia in immunosuppressed people. In healthy individuals, these cause mild colds. In transplant patients or those on long-term steroids, they can lead to hospitalization, lung damage, or death. Routine testing for these viruses is now standard in high-risk groups.
Why don’t immunosuppressed patients have fevers when infected?
Fever is triggered by immune signals like cytokines. When T-cells or other immune cells are suppressed by drugs like steroids or biologics, the body can’t produce these signals. So even a life-threatening infection might cause no fever at all. That’s why doctors rely on tests-not symptoms-to diagnose infections in these patients.
Is Pneumocystis pneumonia only a problem for HIV patients?
No. While it was once called "HIV pneumonia," it’s now more common in transplant recipients and people on long-term steroids or biologics for autoimmune diseases. In fact, in some studies, over half of Pneumocystis cases occur in non-HIV patients. Anyone with T-cell suppression is at risk, regardless of their diagnosis.
Can you prevent infections if you’re on immunosuppressants?
Yes, but not completely. Prophylactic antibiotics (like trimethoprim-sulfamethoxazole for Pneumocystis) and antivirals (like valganciclovir for CMV) reduce risk. Vaccines (flu, pneumococcal, COVID-19) help-but they don’t work as well. Avoiding crowds, wearing masks, and washing hands are critical. Routine screening (blood tests, lung scans, stool tests) is the best defense.
What should I do if I’m on immunosuppressants and feel unwell?
Don’t wait. Call your doctor immediately-even if you feel "just a little off." Say you’re immunosuppressed and ask if you need testing for infections. Request a chest X-ray, blood cultures, or stool test if you have diarrhea. Early testing saves lives. Delaying until you have a fever or cough can be deadly.





SNEHA GUPTA
March 19, 2026 AT 04:09It’s terrifying how silently these infections creep in. No fever. No redness. Just a whisper of fatigue that becomes a scream too late. I’ve seen it in my mother’s post-transplant journey-how doctors dismissed her ‘just being tired’ until her lungs started failing. We weren’t told to test unless she was gasping. That’s the flaw. The system waits for a siren when the warning is a whisper.
What if we shifted from ‘wait for symptoms’ to ‘assume infection until proven otherwise’? Proactive screening isn’t paranoia-it’s precision medicine. We don’t wait for a house to burn before checking the smoke detector.
And yet, most protocols still treat immunosuppressed patients like they’re just ‘sick people.’ They’re not. They’re a different biological category. We need diagnostic frameworks built for their physiology, not adaptations of healthy-person models.
It’s not about more drugs. It’s about rethinking how we define illness itself.
And honestly? The fact that we still rely on CRP and ESR in this population is criminal. Those numbers are meaningless here. We’re using 1950s tools to diagnose 2020s pathology.
Gaurav Kumar
March 20, 2026 AT 17:19India has been dealing with this for decades. Fungal infections in immunocompromised patients? We see it daily in our rural hospitals. But here’s the thing-no one talks about how poverty makes it worse. No access to bronchoscopy? No PCR? No antifungals? You die quietly. And the government? Still thinks ‘vaccines’ are the answer.
Meanwhile, in the US, they’re doing T-cell therapies and metagenomic sequencing. We’re still using chest X-rays and hoping for the best.
It’s not science. It’s inequality dressed up as medicine.
And don’t even get me started on how Western papers act like Pneumocystis is a ‘new’ problem. We’ve been burying patients from it since the 80s. No one cared until it hit rich white people.
🫠
David Robinson
March 21, 2026 AT 09:46Look, I’m not a doctor, but I read the paper. And honestly? This whole thing feels like overkill. I’ve been on prednisone for years. I get a little short of breath sometimes. So what? I breathe deeper. I don’t need a bronchoscopy. You’re scaring people into thinking every sneeze is a death sentence.
And who the hell is paying for all this testing? Insurance won’t cover it. Patients can’t afford it. So now you’re just creating anxiety and bills.
Maybe the real problem isn’t the bugs. It’s the medical industry’s obsession with detecting everything-even when it doesn’t matter.
Also, why is everyone acting like Aspergillus is some new horror? My grandpa had it in the 70s. He died. So did a lot of people. We didn’t have CT scans then. We didn’t have antifungals. We just… accepted it.
Now we’re pathologizing normal human vulnerability. Chill out.
Jeremy Van Veelen
March 23, 2026 AT 06:58Oh. My. GOD.
Have you ever sat in a hospital room watching someone you love turn pale from a fungus that doesn’t even have a name? No? Then you don’t get it.
This isn’t a ‘medical case study.’ This is a horror movie written in lab reports. Pneumocystis doesn’t knock. It *creeps*. It doesn’t scream. It whispers… until your lungs are full of jelly.
And the worst part? The doctors smile. ‘You’re stable.’ They say. ‘No fever.’ They say. ‘Your labs look fine.’ They say.
But your body is being eaten alive. And they’re waiting for you to collapse before they do anything.
I cried when I read this. Not because I’m emotional. Because I’ve lived it.
And now? Now I have to watch my daughter go through the same thing.
They call it ‘immunosuppression.’ I call it a death sentence with a waiting list.
Laura Gabel
March 23, 2026 AT 14:28jerome Reverdy
March 24, 2026 AT 13:30Let’s reframe this: we’re not talking about ‘infections’-we’re talking about *opportunistic reawakenings*. These aren’t new invaders. They’re old roommates who got kicked out when your immune system was strong. Now? They’re back. And they’re not here to chat.
What’s wild is how little we’ve adapted our diagnostic models. We still use ‘fever = infection’ like it’s a universal law. But for these patients, the body’s alarm system is broken. So we’re left guessing.
The real breakthrough isn’t a new drug. It’s the shift from reactive to predictive medicine. We’re starting to do that-genomic screening, serial PCR, AI-assisted imaging. It’s slow. But it’s happening.
And honestly? The fact that we’re even *talking* about HHV-6 in transplant patients 20 years after we first saw it? That’s progress. We’re learning. Slowly. Messily. But we’re learning.
Also-props to the team that started routine BAL testing. That one saved a lot of lives. Small changes. Big impact.
Andrew Mamone
March 25, 2026 AT 11:52Just wanted to say-this is why I became a med tech. I worked in a transplant unit for 5 years. Saw a kid with no fever, no cough, just lethargy. BAL came back positive for Pneumocystis. He was 12. He didn’t even know he was sick.
Got him on TMP-SMX. Added steroids. He went home in 10 days.
That’s the moment I realized: medicine isn’t about fixing what’s broken. It’s about catching it before it breaks.
❤️
MALYN RICABLANCA
March 26, 2026 AT 08:11OMG. OMG. OMG.
Did you know that CMV can cause *bone marrow failure*? Like… your body just… stops making blood? And then you get sepsis from having zero white cells? And then you get fungal pneumonia? And then you die? And no one knows why? Because you didn’t have a fever?!
And they say ‘just get tested’? But the test costs $1,200 and your insurance says ‘not medically necessary’? And your doctor says ‘maybe it’s just stress’? And you’re left wondering if your next yawn is your last?
I’ve been on tacrolimus for 11 years. I’ve had 3 hospitalizations. All from ‘silent’ infections. All missed. All almost fatal.
And now? Now I have to pay $800 a month for prophylactic meds that ‘might’ help. And my employer’s insurance says ‘you’re high-risk so we’re dropping you.’
THIS IS A HUMAN RIGHTS ISSUE.
WHO IS MAKING MONEY OFF THIS? WHO IS PROFITING FROM OUR SILENT SUFFERING?
WHERE IS THE OUTRAGE?
…I’m crying.
😭😭😭
gemeika hernandez
March 26, 2026 AT 18:26Nicole Blain
March 26, 2026 AT 20:17This is why I always ask my oncologist for the ‘silent infection panel’ now. I don’t wait. I don’t hope. I ask. ‘Can we check for Aspergillus? Pneumocystis? HHV-6?’
They roll their eyes. But they do it.
And last month? We caught it early. One test. One antifungal. No hospital.
So yeah. Ask. Even if it feels silly. Even if you’re ‘just tired.’
It’s not paranoia. It’s self-care.
🌸
Kathy Underhill
March 27, 2026 AT 18:24The most important line in this whole post: ‘Normal doesn’t exist.’
We’ve built a medical system based on averages. But immunosuppressed patients aren’t averages. They’re outliers. And outliers get ignored until they die.
What we need isn’t more tests. It’s a new framework. One that doesn’t assume ‘healthy baseline’ as the norm. One that says: ‘This person’s body works differently. So we treat it differently.’
It’s not radical. It’s basic human dignity.
And yes-prophylaxis, screening, and early intervention save lives. Not because they’re flashy. Because they’re thoughtful.
Srividhya Srinivasan
March 27, 2026 AT 22:20I knew it. I KNEW IT. This is all from the vaccines. The biologics. The ‘immunomodulators’-they’re not fixing you. They’re *unmaking* you. The government knows. The pharma companies know. That’s why they push all this testing-so you think you’re safe. But you’re not. You’re being slowly hollowed out.
And now they want you to pay for PCR tests? To prove you’re dying from a ‘silent infection’? That’s not medicine. That’s a trap.
My neighbor got a transplant. Now she’s on 17 pills a day. She can’t go outside. She can’t hug her grandchildren. She’s terrified of the air.
Who benefits? The labs. The hospitals. The drug companies.
And you? You’re just a data point.
Don’t trust the system.
Protect yourself. Stay home. Eat garlic. Pray.
They’re not saving you. They’re monetizing your fear.
Prathamesh Ghodke
March 28, 2026 AT 15:13Man, this hits different when you’ve sat with someone who died because no one thought ‘shortness of breath’ was serious.
My uncle was on prednisone for lupus. Got a little winded. Said ‘I’m just out of shape.’ Doctor said ‘keep exercising.’ Two weeks later, he was gone. Pneumocystis. No fever. No cough. Just… gone.
So now? I tell everyone I know: if you’re on immunosuppressants, and you feel *anything* off-push for a BAL. Push for a stool PCR. Push for a CMV load.
Don’t be polite. Don’t be ‘nice.’ Be relentless.
And if your doctor brushes you off? Get a new one.
It’s not drama. It’s survival.
And yeah-I’ve sent this to 3 people today. One of them just got tested. It was positive. Early. They’re on meds now.
Small wins. They matter.
Stephen Habegger
March 30, 2026 AT 09:09Just wanted to say-this is why I love modern medicine. Not because it’s perfect. But because we’re *trying*. We’re not ignoring the silent killers anymore. We’re building tools to find them. And that’s huge.
Yes, it’s expensive. Yes, it’s uneven. But we’re moving.
And every time someone speaks up-like this post-it helps. It pushes. It changes.
Keep talking. Keep asking. Keep pushing.
You’re not alone.
💪
Justin Archuletta
March 30, 2026 AT 13:53